In today’s world, health is a fundamental value and the highest priority for every individual, and we understand that each body has its own unique language. This language is not always easy to interpret.
However, the functional diagnostic medical equipment produced by our company makes it possible to hear this language.
On June 9, 1999, the Ukrainian company “DX-SYSTEMS” was founded to produce medical diagnostic equipment. Since then, it has been steadily growing, guided by its own philosophy, mission, goals, and ambitions.
Our slogan, “The body speaks – we translate!”, symbolizes our deep understanding of the importance of precise measurements and reliable data in medical diagnostics and scientific research.
Our medical equipment transforms complex physiological signals into accurate and understandable data, which are crucial for comprehensive analysis, deeper insights into a patient’s health, and confident diagnosis.
By creating such equipment, we aim to expand the boundaries of diagnostics, providing healthcare professionals with more data, precise measurements, and detailed results, thereby opening up greater opportunities for informed decision-making.
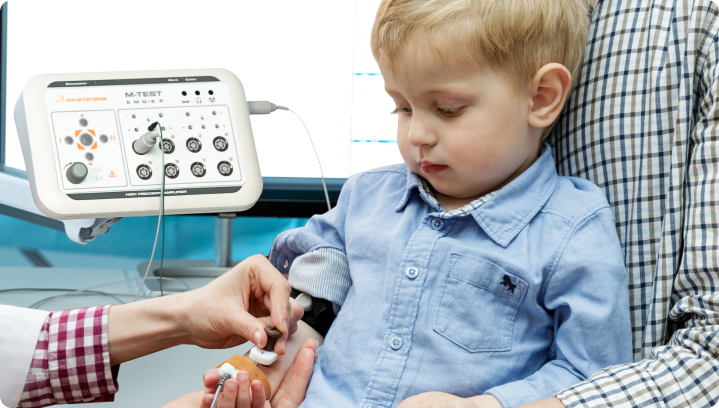
We create medical devices that translate body’s signals into accurate data
On June 9, 1999, the Ukrainian company “DX-SYSTEMS” was founded to produce medical diagnostic equipment. Since then, it has been steadily growing, guided by its own philosophy, mission, goals, and ambitions.
Our slogan, “The body speaks – we translate!”, symbolizes our deep understanding of the importance of precise measurements and reliable data in medical diagnostics and scientific research.
Our medical equipment transforms complex physiological signals into accurate and understandable data, which are crucial for comprehensive analysis, deeper insights into a patient’s health, and confident diagnosis.
By creating such equipment, we aim to expand the boundaries of diagnostics, providing healthcare professionals with more data, precise measurements, and detailed results, thereby opening up greater opportunities for informed decision-making.
Creation of a series of hardware-software systems for electroencephalography (BRAINTEST), rheography (RHEOTEST), and electrocardiography (CARDIOTEST) based on Windows.
Initiation of the development of a new line of equipment – electronic neuromyographs of the M-TEST series.
Migration of medical devices BRAINTEST, RHEOTEST, and CARDIOTEST to PC connection via USB port and development of new amplifiers.
Development of M-TEST electronic neuromyographs – expansion of functional capabilities, hardware improvements, and software updates.
Next generation EEG: amplifier enhancement, development of a new VIDEO EEG module – expanding software capabilities, and creation of new EEG accessories.
Second generation of M-TEST electronic neuromyographs: development of a new amplifier and stimulator, expansion of software features, refinement of stimulation protocols, and creation of the “Chewing Test” mode.
Migration of all serially produced devices to a new-generation component base, significantly improving the technical performance of the equipment.
Enhancement of all produced devices.
Complete modernization of the electrocardiograph.
Development of the “Nystamography” mode in the M-TEST series devices.
Creation of English-language versions of BRAINTEST, RHEOTEST, and CARDIOTEST.
Development of Bluetooth-enabled ECG.
Migration of all software projects to a new development environment: from Visual Studio 2010 to 2015, and later to 2018.
Third generation of M-TEST ONE electronic neuromyographs: development of a unified amplifier device combining a current stimulator, photo- and phonostimulator; software enhancements (motor inching, rhythmic stimulation according to scenario, “Registration” window).
Complete modernization of BRAINTEST EEG: new amplifier, FSS, accessories, and additional EEG analysis functions – “Amplitude Analysis” mode.
Development and refinement of the next-generation BRAINTEST PLUS EEG devices.
Development of a new M-TEST.NET software platform.
Creation of a new M-TEST ONE electronic neuromyograph with improved technical specifications.

Our medical devices are trusted by a wide range of specialists, including neurologists, neurosurgeons, functional diagnostics experts, sports medicine practitioners, and rehabilitation professionals. They are also valued by researchers and educators in medical institutions, supporting advanced diagnostics, treatment, and education.
At the national level, we hold stable positions among the leading manufacturers of medical equipment for functional diagnostics.
DX-SYSTEMS is the only manufacturer of electroneuromyographs in Ukraine and one of the few in the world. We have a wide network of partners, including hospitals (state, municipal and private), medical centers, educational institutions, etc.
The international presence of our company is an important element of the development strategy, which allows us not only to increase sales volumes, but also to contribute to the development of world medicine. This opens up new prospects for us and helps us gain international recognition.

Our strength lies in our team – a group of like-minded professionals united by a common goal: to create high-quality medical equipment.
Each of us contributes uniquely to this process, combining expertise, creativity, and a drive for continuous improvement. We work together as a single, well-coordinated mechanism, supporting one another at every stage. We believe that true success is achieved only through collaborative teamwork and joint effort.
Every day, we focus on developing and upgrading our equipment, paying attention not only to technological processes but also to ease of use – an essential factor for medical professionals.
In designing and refining medical devices, we constantly listen to the needs and feedback of physicians, as their daily practice and experience serve as our source of inspiration and development. Every new device or improved model of an existing one is born from the real needs of healthcare professionals, aimed at making their work easier and creating a reliable assistant – not just a tool, but a powerful link between doctor and patient.
We believe our medical equipment helps doctors see more, diagnose more accurately, and respond faster. Hear the language of the body!

CEO

CFO

CTO
Our company has developed, implemented, and certified a robust quality management system in full compliance with DSTU EN ISO 13485:2018 (EN ISO 13485:2016, IDT; ISO 13485:2016, IDT) – “Medical devices. Quality management system. Regulatory requirements.”
This certification reflects our commitment to producing reliable, high-quality medical devices that meet both national and international standards.
Our medical devices fully meet national and international standards, including the Technical Regulations on Medical Devices (Ukraine) and the EU Council Directive 93/42/EEC, in line with the transitional provisions of EU Regulation 2017/745. This ensures that our equipment is safe, reliable, and suitable for use in healthcare settings worldwide.